Introduction:
Active surveillance (AS) is the currently the recommended treatment for very low- and low-risk prostate cancer. AS is also being more frequently used for favorable intermediate risk prostate cancer in select patients. Clinical tools such as prostate multiparametric MRI (mpMRI) and genomic classifiers such as Decipher® GC are recommended to risk stratify newly diagnosed prostate cancer (PCa) patients in the pre-treatment setting.
Our aim for this study was to examine the relationship between pre-treatment parameters such as PSA, biopsy Gleason Grade Group (GGG), MRI Prostate Imaging-Reporting and Data System (PI-RADS) score and Decipher® GC, and progression to treatment in patients currently on AS for PCa.
Methods:
We retrospectively examined patients at our institution who underwent Decipher® GC testing on prostate biopsy tissue (n=888) between December 2016 and March 2023. Data regarding PSA, PI-RADS lesion on MRI, biopsy GGG, Decipher® GC and treatment modality were collected. Of these patients, 235 were placed on AS initially. Ultimately, 88 patients progressed to treatment. We performed a multivariable Cox proportional hazards model with time to treatment as the outcome and PSA, PI-RADS score and Decipher® GC as potential predictors.
Results:
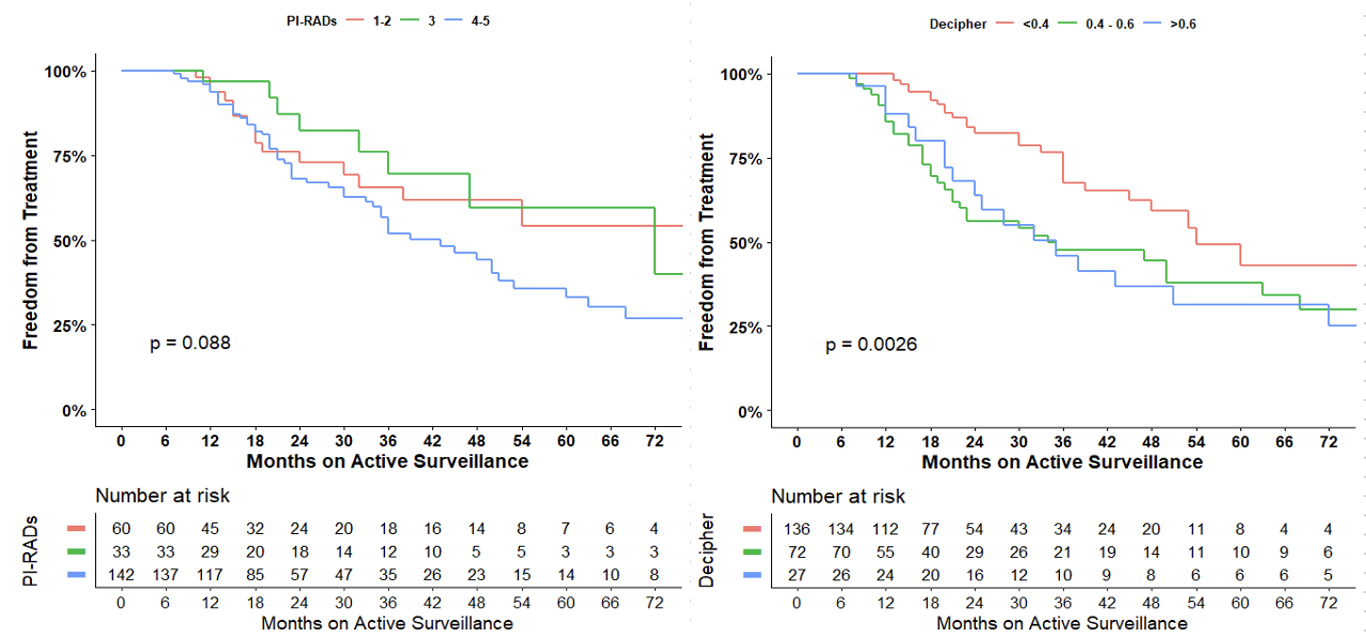
Our final cohort included 235 patients initially placed on AS. The cohort had a mean PSA of 7.4 and Decipher® GC score of 0.38. 25.5% of these patients (n = 60) had PIRADS 1-2 lesions on MRI, 14% (n = 33) had PIRADS 3 lesions and 60.4% (n = 142) had PIRADS 4-5 lesions. 40% of these patients (n = 94) had GGG1, 48.1% (n = 113) had GGG2, and the rest (n = 28) had GGG3 or higher on biopsy.
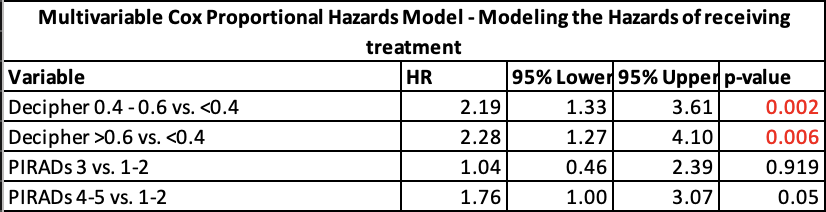
We found that a Decipher® GC above 0.4 (p = 0.002) and 0.6 (p = 0.006) was associated with progression to treatment while on AS, whereas having a PI-RADS 3 lesion (p = 0.92) and having a PI-RADS 4-5 lesion (p = 0.05) were not (Table 1). Survival curves showed that increased Decipher® GC was associated with decreased freedom from treatment, whereas PI-RADS score was not (Table 2).
Conclusion:
Tumor-based molecular assays such as Decipher® GC are included in the NCCN guidelines for localized prostate cancer management. Decipher® GC has been shown to independently predict adverse pathology with ver low-, low- and intermediate-risk PCa. Previous studies have shown the association of Decipher® GC score with progression to treatment on AS without incorporating mpMRI data. We found that an increased Decipher® GC is an independent predictor of progression to treatment in a cohort of patients on AS, while mpMRI (PI-RADS) is not.
Funding: N/A
Image(s) (click to enlarge):
Decipher® Genomic Classifier Score on Initial Biopsy is Associated with Progression from Active Surveillance to Treatment in Prostate Cancer
Category
Prostate Cancer > Potentially Localized
Description
Poster #237
Friday, December 1
1:45 p.m. - 2:45 p.m.
Presented By: John Sheng
Authors:
John Sheng
Joel Vetter
Nimrod Barashi
James Riley McGinnis
Eric Kim