Introduction:
The AUA/SUO guidelines for upper tract urothelial carcinoma (UTUC) recommend endoscopic evaluation as part of the diagnostic workup with grade of tissue as the driving factor for risk stratification. In addition to being a risk factor for intravesical recurrence, ureteroscopic biopsy has a poor concordance with final pathologic stage. Because of this, patients and clinicians must make uninformed decisions regarding various aspect of treatment such as the use of neoadjuvant chemotherapy and intraoperative lymph node dissection. As such, we sought to create a genomic model that was predictive of stage in patients with UTUC.
Methods:
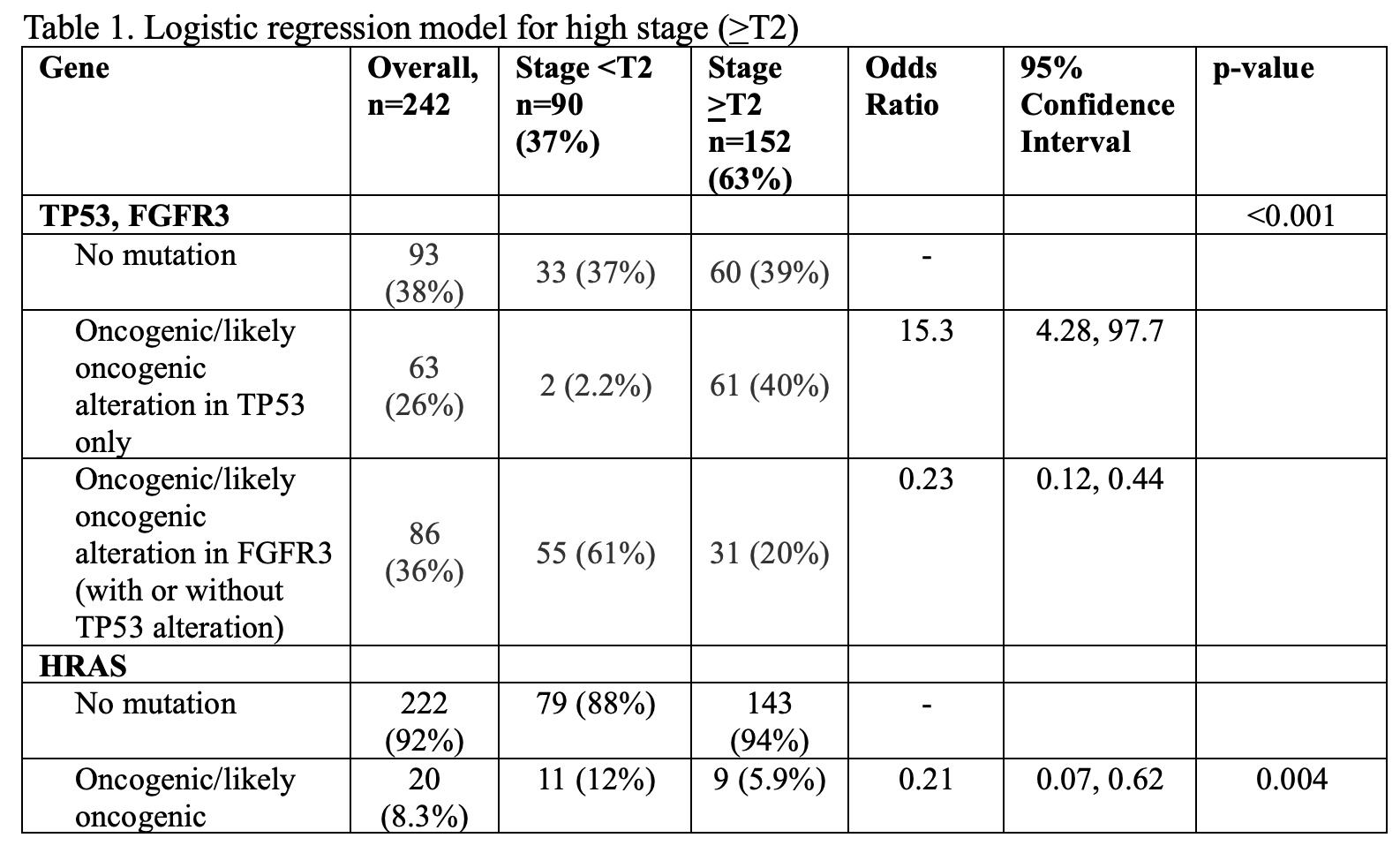
A retrospective cohort of patients that underwent nephroureterectomy and next generation sequencing with MSK-IMPACT, a targeted sequencing panel covering up to 505 genes were included. Diagnoses were dichotomized into low (<T2) or high (>T2) stage based on final pathology. Genes mutated with >5% prevalence and oncogenic or likely oncogenic based on OncoKB annotation were considered for inclusion. Five-fold cross validation was used to compare the following logistic regression models: (1) including only known predictors of stage, TP53 and FGFR3; (2) including only genes significant in univariable analyses after adjustment for TP53 and FGFR3; and (3) logistic regression with stepwise selection. The median and interquartile range (IQR) for sensitivity, specificity, and the area under the curve (AUC) are reported, where sensitivity refers to the proportion of high stage tumors that were predicted to be high stage, and specificity refers to the proportion of low stage tumors that were predicted to be low stage.
Results:
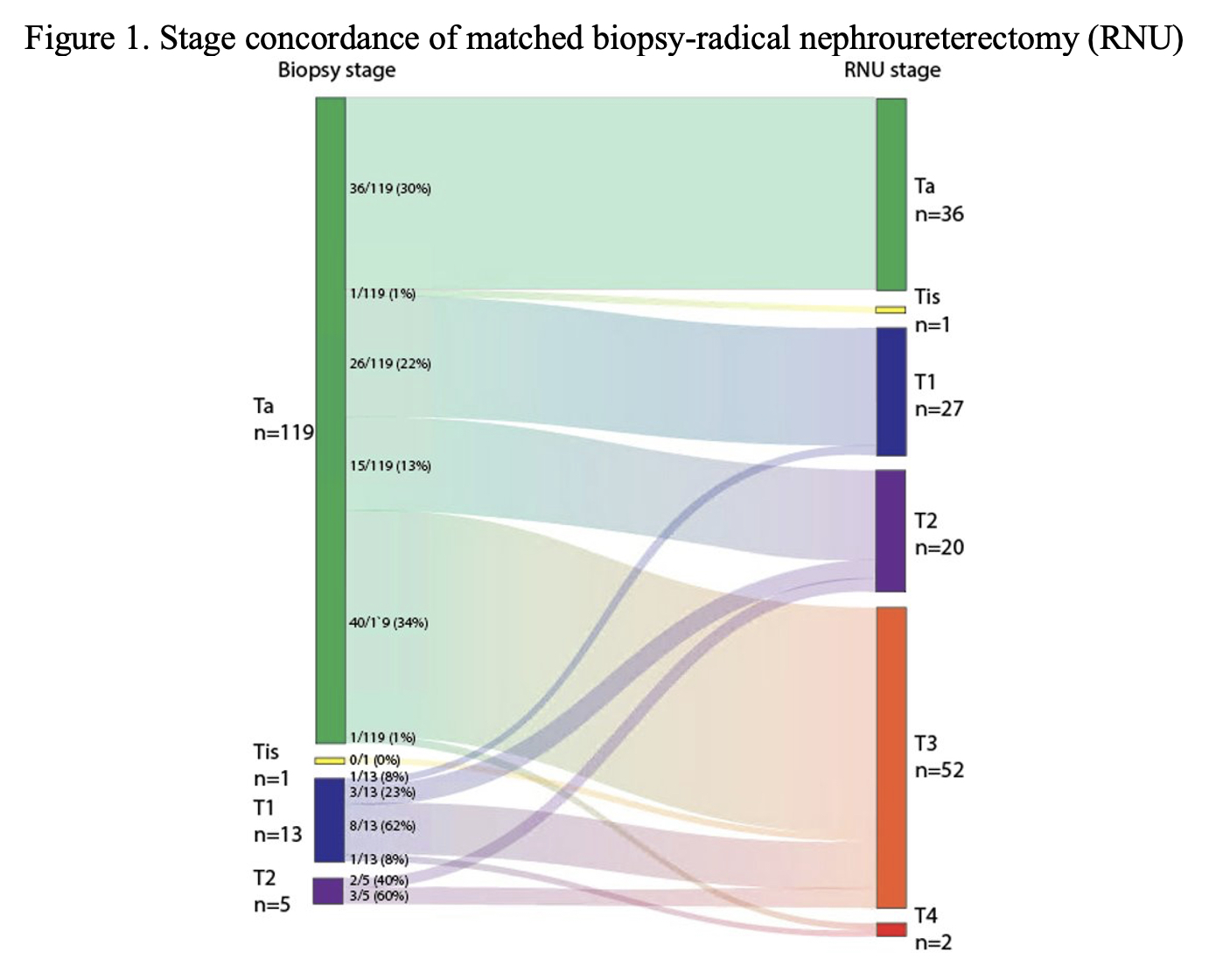
242 chemotherapy naïve patients were included in the discovery cohort, of which 152 (63%) were stage >T2. Stage based on biopsy was only 32% concordant with pathologic stage, with 7% sensitivity and 100% specificity for distinguishing high versus low stage disease (Figure 1). HRAS mutations were significantly associated with increased likelihood of low stage disease after adjustment for TP53 and FGFR3. The multivariable logistic regression model with TP53, FGFR3, and HRAS was selected as the optimal model. Using these three genes in a logistic model resulted in a median AUC of 0.79 (IQR 0.76-0.85). The sensitivity and specificity of the model were 0.60 (IQR 0.50-0.70) and 0.78 (IQR 0.74-0.83), respectively.
Conclusion:
We developed a three-gene model that was predictive of high stage disease. Our model significantly outperforms the accuracy of uretereoscopic biopsy in a development set but is yet to be validated in this population. Ultimately, the clinical utility of this model may be seen not only in improving ureteroscopic biopsy accuracy, but also as applied to sequenced urinary tumor DNA as a non-invasive biomarker. The candidate model is supported by previously published feasibility and concordance studies; validation of this prediction model is planned.
Funding: N/A
Image(s) (click to enlarge):
DEVELOPMENT OF A GENOMIC PREDICTION MODEL FOR UPPER TRACT UROTHELIAL CARCINOMA STAGE
Category
Bladder Cancer > Upper Tract Urothelial Carcinoma
Description
Poster #48
Thursday, November 30
8:00 a.m. - 9:00 a.m.
Presented By: Andrew B. Katims
Authors:
Andrew B. Katims
Jessica A. Lavery
Irina Ostrovnaya
Bernard H. Bochner
Guido Dalbagni
Timothy A. Donahue
S. Machele Donat
Harry W. Herr
Eugene J. Pietzak
Hikmat Al-Ahmadie
David B. Solit
Jonathan A. Coleman